|
It's been an insanely busy term as I'm sure many will appreciate but it's time to share the latest lab news! Starting with papers I have to say how pleased I am for Olivia who's first paper is now online. She's worked incredibly hard and it's paid off with some really nice findings! The paper outlines how two wheat lines respond to steady or variable water availability under low or high nitrogen. It's one of the first projects to use the drought spotter at the Australian Plant Phenomics Facility at University of Adelaide which automatically waters individual pots based on weight and as frequently as programmed. This paper is soon to be followed with the next set of exciting results! Keep an eye on her! More good stuff to come! Also on the paper front, our paper describing the use of treasure hunt mobile software for undergraduate education is also now online in Plants People Planet. I'll also be presenting this work at SEB in July and as a poster at Plant Biology in August so do come and chat to me about it! Excitingly our paper combining microCT root imaging with microdialysis nutrient analysis with Richard Brackin from the University of Queensland is a top 20 paper in Plant Cell and Environment. Thanks to Richard for his help on this project during his visit and thanks to Rank Prize Funds New Lecturer Award for the funding for this work. The team has also had success recently at the Plant Environmental Physiology Group Early Career Event which was held at Eden Project this May. Magda and Findi both attended, meeting other PhD students and postdocs working in different areas of plant physiology. Magda took home the Poster prize and together with the talk winners wrote a really nice blog about the event. Do follow PEPG on twitter for updates on events (@PEPG_SIG). Also still on my awesome team: Daisy is currently on work placement with BBC Sky at Night and you can read her articles here. It's a fantastic opportunity as part of her BBSRC DTP studentship but all organised by her! I'm looking forward to hearing all about it when she returns!
The semester has been filled with teaching. I was flattered by some very high teaching scores for my third year class. Equally I was happy with the scores from my first year class which is a large class, a compulsory module for many, and faced some administration challenges at the start of term. Adding to that I'm female and not a professor so I was pleased with teaching scores in line with the school average. To be honest the comments are more useful than any metric and supported my suspicions about things that could be better and those that already work well. Perhaps one of the most fun teaching activities this semester was the first year plant science revision session. I designed this based on the BBC2 Eggheads gameshow. The lecturers from the module made up the Egghead team and 5 students volunteered to be the challenger team. Just like on BBC2 there are themes (in this case based on the lecture topics) and a series of one-on-one question rounds based on multiple choice questions. The challenger team can choose which academic to take on for each one-on-one round and the winners of each round could join their team for the final general knowledge round in which the teams could confer before answering. To increase audience engagement I added an ask-the-audience option using Mentimeter. I was pleased to see about half the class attended the session (110 in the class) even though it was the only teaching week after Easter before the exams meaning for many that was the only lecture that week. I was really impressed with the students on the challenger team who all did brilliantly! They won every round and were only stumped on one question in the final round! I also want to acknowledge the academics for participating - it's quite nerve wracking being put on the spot for topics they didn't teach. All in all it was a lot of fun and I hope the students found it a more interesting way to revise - and a way to get hold of any of the academics to answer specific questions before the exam! Finally the summer is almost here! This conference season I'll be at SEB in Seville and special thanks to ASPB for the Sharon Gray Women's Young Investigator Travel Award to attend ASPB Plant Biology in San Jose - very much looking forward to those. I'm also super excited to be visiting Erin Sparks for much of the summer. Although we have been Skyping monthly, we have never met in person. To those critical of Twitter - this collaboration is purely a result of the great opportunities that Twitter offers! I'm very much looking forward to working with the rest of her team on physiology and seeing their mechanical field trials in practice! I will do my best to write a science travel blog while away so watch this space over the coming months! I think that's enough for now! There is a new choose-your-own-adventure story for Eca coming soon so watch out for that in the coming few days! Latest papers: Hartman T, Lydon SJ, Rasmussen A (2019) Hunting for Answers: Linking lectures with the real world using mobile treasure hunt app Plants People Planet; http://doi.org/10.1002/ppp3.33. Cousins OH, Garnett TP, Rasmussen A, Mooney SJ, Smernik RJ, Cavagnaro TR (2019) To stress or not to stress: Plant and soil responses to variable water and nitrogen supply. Plant Science. (http://doi.org/10.1016/j.plantsci.2019.05.009)
1 Comment
It’s been a while since my last blog - travel, writing, teaching - lots of material for future blogs, but today I want to share a green adventure from this weekend and a botanical first for me! Three times a year the Hillier-Lancaster Plant group brings together an amazing array of people from all aspects of the botanical world. This meeting’s adventure, exploring the Sir Harold Hillier Gardens, found our heady mix of obsessive plant geeks soaking up the history of the Hillier family legacy while chatting about plants that take our fancy. The ebb and flow of conversations and discussions are punctuated with stories by the phenomenal Roy Lancaster. Stories of the origins of particular plants growing in the gardens and what they mean to him and others are inspirational. My favourite was a story about the first time he learnt a particular tree that he had seen tantalisingly from behind a wall, and he described the excitement of learning what it was. He encourages us to remember that first time we encounter different plants and likens it to a first kiss – not necessarily the person you want to be with forever, but something never to be forgotten. I love this description of that excitement and curiosity and I encourage everyone to share in this joy and pass it onto others. For me the stories behind the plant are as exciting and important as the plant species or why it survives where it is. The adventures others went on to bring that seed to that place or the events linked to the variety or species are equally inspiring – it’s like living through the plants! As a nobody from the back of nowhere, growing up below the poverty line, this has always been part of my passion for plants. Reading about the early botanical-explorers in Australia – and elsewhere – planted a seed (full pun intended) that perhaps I could ‘go somewhere’ – maybe not as a physical explorer but as a scientific one. I’m still a nobody, but I am having scientific adventures and exploring the frontiers of plant physiology. There are still challenges and hurdles as a female, foreign, non-middle class person, but recently living through plants has taken a new meaning for me. At the age of 34 my confidence took a blow when the signalling in my heart failed requiring a pacemaker to correct. Relearning who I am, and what makes me tick (literally and figuratively!) is a slow process, and 3 years on, the road to recovery struggles on. But moments like those from this weekend remind me of the adventures and the excitement that continues. Remind me that at my core is still a plant geek that chases weird plants (like the Amorphophallus in flower at Wisley - below). These amazing people remind me it’s ok to spend weekends doing something I love that also makes up my job. That it’s ok to have a blurry line between work and play. That I never want to forget the excitement of ‘first time’ for each plant! And I encourage others to go out as spring springs and meet a new plant and share that excitement of the first time! A first time for me today: Amorphophallus at Wisley (Left). I'd love to say the queue was for this beauty but actually not one person (except me) noticed it amongst the lego animals (possibly a blog for another day) - but somehow lining up to tick this plant off my list seems appropriate!
As we come to the end of another academic year and people debate whether we are giving out too many firsts, I find myself pondering a few questions.
But before I start people arguing I should make clear that I completely agree that we need to ensure a level of consistency for our students. Especially in the light of exceptional circumstances like snow interruptions to lectures and practicals which don't happen to every cohort. Ok so that said I think one of the things we need to decide as lecturers is:
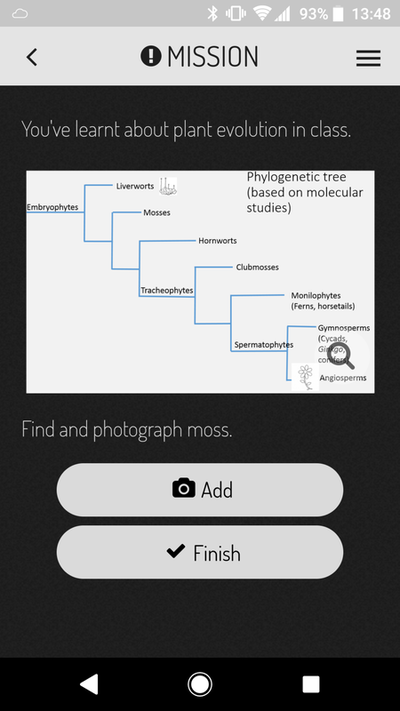
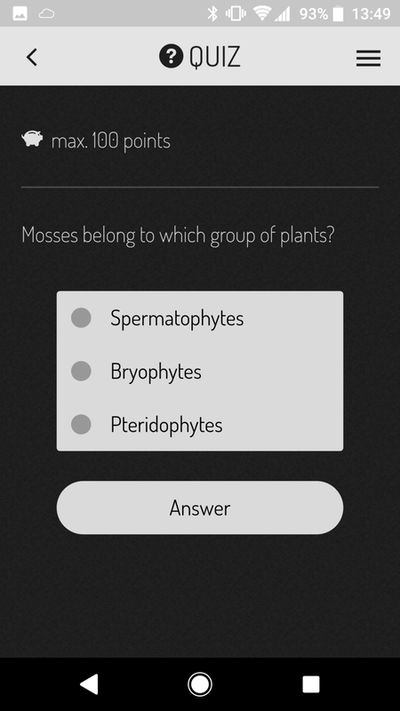
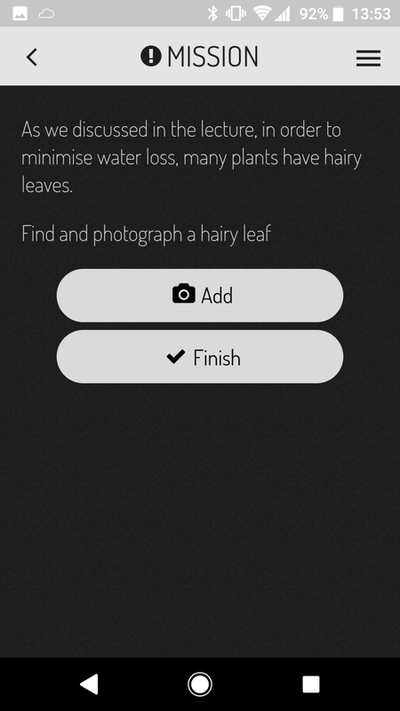
Is it to teach a specific set of skills (or knowledge) to our students? Or is it to produce a clear ranking of who's the best and who's the worst? This question lies at the heart of the debate because the two goals require different teaching and evaluation strategies. If we are providing our students with skills and knowledge then as good teachers we should be providing all the students with the best opportunity to learn those skills and knowledge. Good teaching strategies will reach diverse students, enhancing their learning through safe and motivating learning experiences. Under this scenario, in theory, all students should have the chance to achieve high grades if they participate in those learning experiences and acquire those skills. In addition, under this scenario since we are good teachers we will continue to adapt our teaching strategies to enhance student learning from year to year which should result in more students acquiring the skills and knowledge in subsequent years, increasing the number of firsts (of course as someone who cares about teaching, I'd love to give the credit of rising grades to the teachers!). But should we stop adapting once we have the perfect way to impart those skills? This brings me to the next point - students are starting uni with different skills. Similarly the jobs we are training them for also have evolving expectations. So in an ideal world we should be adapting our learning goals to bridge between entry skills and job requirements. A bit obvious? Ok well if we increase our learning expectations from year to year how can we compare a first from this year to a first in 5 or 10 years time since the expectations will have changed? I think this will be very important for us to consider when comparing cohorts in the near future as I would argue we are in a transition period from memorising knowledge to skills acquisition - in particular complex problem solving and team/people skills (to name a few - a topic for another blog). If we explore the other option where our teaching goals are to rank the students then what does this mean to employers? Students have so many options especially in their final years - each of which teach a different set of skills or knowledge - a student with a first may still not be the best option for a particular job if they had selected modules more suited to a different job…. My personal philosophy (as you may have guessed from the biased way this is written!) is that we are here to teach skills and if the students learn those skills they should all have the opportunity to get high grades. I also feel that the skills required for success in a changing world are different to what they were in the past. Why am I pondering all this? In exam board there was a point made about a new aspect of assessment that I introduced to my third year module which resulted in good spread of grades. However that wasn't my intention when I instigated the change. In the past I had noticed that third year students still find writing concisely a challenge. In response to this observation the final coursework (two short essays on topics linked to the lectures) included two marking criteria. The first was the standard criteria they have been evaluated against their whole degree. The second was worth 25% of the grade and was deliberately written to evaluate concise writing skills. To ensure the students had the best chance to learn these skills I ran a formative workshop where students submitted a paragraph of text the night before and during the session I projected the paragraph on the screen (anonymised) and as a group (there are 13 in the class) we reworded the paragraphs to be more concise without losing the meaning. I provided a handout with a substitution table which contained common long-winded statements and concise alternatives. The marking criteria included use of the substitution table along with a few other points and we worked through the new marking criteria in the workshop so the students knew exactly what was expected. I confess when marking some of the essays I was disappointed that although overall there was good improvement, there was still fluff in some of the essays. This is what resulted in the mark spread for which I was complemented during exam board. I actually felt this was a failing of my teaching - that there is more I can do to enhance these skills next year. But is spread a bad thing? Maybe some spread is a good thing? It certainly shows we are stretching the students with skills not already learnt. The students expressed appreciation for the skills training (in person, in the module evaluation and to the examiner who asked). I will still strive to improve writing skills further next year which may result in less spread…I wonder what will be said at exam board if one year there is good spread and the next year there is less!? Do I have a point to this blog? Not really except to prompt further thought on firsts, spread, comparing cohorts and what is our underlying goal of undergraduate teaching. So what do you think? APPlying theory to realityIt was a bleak Wednesday morning, rain sheeting down diagonally from the blistering cold wind. Sitting in the back of my half empty class while a guest lecturer has the floor, daydreaming of warmer climes. The prospect of collecting plant material (my afternoon job) in these dismal conditions not filling me with joy. I check my weather app for the 4th time in as many minutes….it hasn't changed and for that I'm grateful, although doubtful. 'Clearing by 1 pm' looks less likely as the morning drags on. More concerning for me is the weather for Thursday when I take first years outside to learn about plant traits. Blissfully the forecast is correct and the rain stops. Even more amazing, Thursday dawns a stunning clear sunny day that's almost warm (says an Australian). The practical is for a first year module called Life on Earth with 200+ students from a diverse range of programs including biology, biochemistry and environmental sciences based on our University Park campus. From my (geeky) perspective this provides a captive audience with whom to share some of the cool things about plants! The first plant practical is my responsibility and I designed the session around observational skills - important no matter what degree program is being studied - and peer learning with students working in groups of four for the duration of the session. At the core of the activity sequence is a mobile phone app - ActionBound - selected for ease of use and diverse phone compatibility. The learning sequence was organised in three stages:
The app is little bit like a roaming Rogo exam (which they have for every other practical but without any negative associations). Students received all the marks (5%) for the practical by engaging with the session and I emphasised that making mistakes is perfectly ok to encourage a safe learning environment. Mistakes made are far more likely to be remembered next time they come across the information. I also ran a similar activity in the first year Plant Science module on our Sutton Bonington campus with modifications related to the paper-less/mobile-less new teaching lab. Stage one and two are based on an activity from BIOL1030 at The University of Queensland where I was a demonstrator from 2008-2010 and run by Louise Kuchel and Robbie Wilson (back then). In this case the plant key was made in the lab and several weeks later students were taken on a field trip to the Rainforest where they used their key on plants labelled along the walking track. There are many great ideas being applied in that module and I give full credit to them for the inspirational ideas. Back to Nottingham: Before the students arrive in the lab, plant samples collected the day before and stored in the cold room need arranging on benches. In the process of setting up I discover that half of the samples of plant 6 have disappeared. There must be a pile of nicely labelled stems sitting out by the lake somewhere! I blamed the squirrels and the groups missing plant 6 were encouraged to use the process of elimination to work out which letter labelled on an outside tree would correspond to plant 6 in the lab. As the students get under way I'm quickly aware of the biggest problem with the activity - and its not the activity. Since we have ethics approval to evaluate the effectiveness of the activity students need to sign a consent form which Susie or myself need to counter sign. This was a massive bottleneck and meant that instead of being able to interact and talk with the students I was signing forms for the first hour - this must change next year. Once the forest of paper was signed, we headed out into the glorious sunshine by the lake where the outdoor stages took place. From a teaching perspective it was a lot of fun roaming around the trees talking to the students providing extra tid-bits of information about plants in a low-stress environment. In the 90 minutes or so we were outside I think I spoke to most of the 50 groups at least once - all actively seeking the plant features for the treasure hunt and all of them engaged in conversations with me about wider topics.
At the end of the treasure hunt there were a few questions about their experience and once they clicked 'send' the responses become visible on my laptop. This meant I could send a document to all the students in the following days to clarify common misconceptions with additional information. In the evening after the prac, I opened an anonymous survey with questions about their experience, favourite question types and any other comments. I now have a lot of data to sort through but here's a taster from the students:
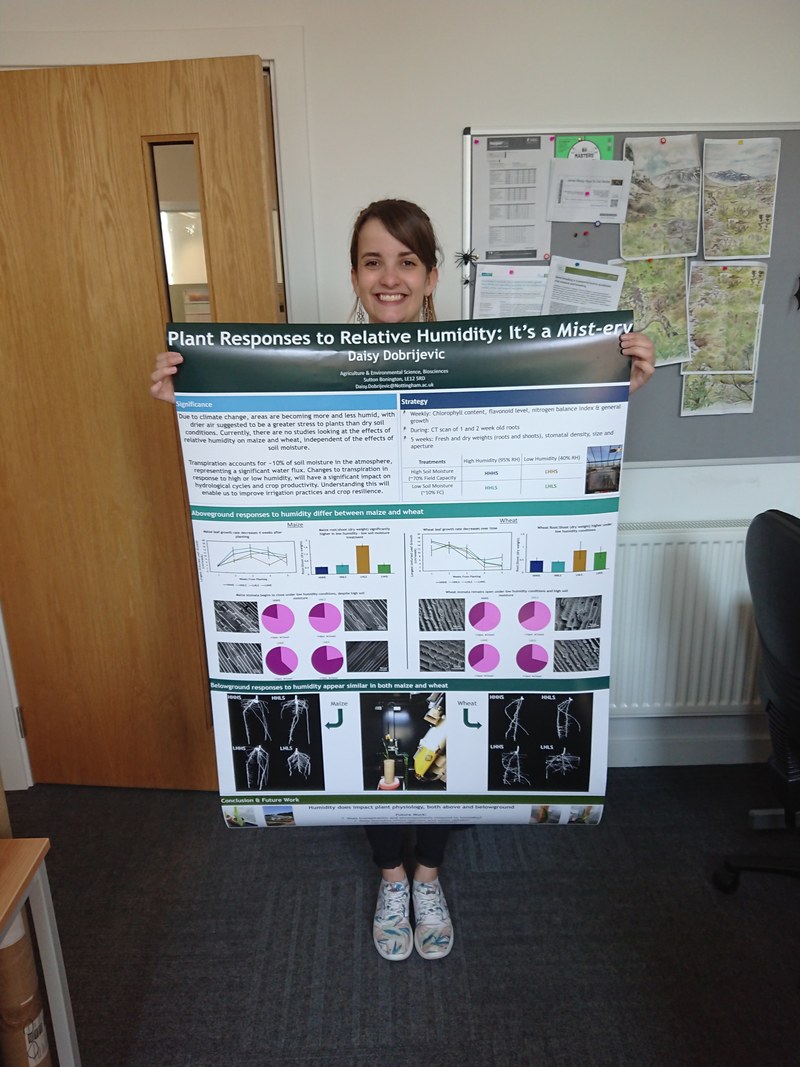
Many of the students also made suggestions on improvements and all of those who took the time to provide suggestions gave positive responses to how the activity helped them learn. Once the data has been sorted the next round of tweaks will begin, after all teaching is an iterative process of implementing, evaluating, and adapting - just like the scientific process. I also think there's a lot to be said for the mental health benefits of being out in the fresh air and sunshine in a low pressure learning environment. A topic to be explored another day. Thinking back to that bleak Wednesday morning, I needn't have worried and the irony in my lack of trusting weather apps when I'm willing to trust apps in my teaching, is not lost on me! I'd also like to take the opportunity to thank Susannah Lydon for her help, support and signatures! I'd also like to thank the support team who help every week with preparing material for practicals both at Sutton Bonington (led by Darren Hepworth) and at University Park (led by Mike Gubbins). Without them, none of this would be possible. I'd also like to thank the team of demonstrators who helped out on the day and finally thanks to all the students who engaged in the activity and provided their perspectives in the survey. :) Thank you. Just a quick post to congratulate Daisy Dobrijevic for adding the BBSRC DTP spring school poster prize to her Gold for the AES 1st year talk at the postgrad symposium last week! She's keen to go into science communications after her PhD ....coming to a tv channel near you! You can check out her podcast here on the UoN Radio.
Well done Daisy! :) At a time filled with personal and professional challenges one of the things that never fails to bring me joy is my team. Last week all six of my students (yes I even claim the ones I co-supervise!) made me very happy (is proud too patronising?).  Sutton Bonington has a campus-wide postgrad symposium at which first and third years give a 10 minute talk, the third years also chair sessions and the second years present posters. All students are expected to participate which meant the whole team was in one place for the first time, with Magda and Darwin travelling in from NIAB-EMR and Rothamsted, and Olivia having just arrived back from 2 years in Australia. Unfortunately I had to miss the first day of the symposium talks (teaching conference) and so missed Olivia's third year talk, but by all accounts she did very well. Of particular note another academic made a point of telling me how well she chaired a particularly challenging session at the end of the Thursday program (an achievement, that she admitted, had worn her out!). This is quite an honour. I definitely find chairing sessions harder than any other form of presenting! Not only do you have to speak in front of the audience, you have to keep time (a feat in itself sometimes) and listen carefully enough to think of sensible questions under pressure! (perhaps future post-grad symposiums could include a chairing award!). When I arrived on campus to take everyone for dinner, our team were milling around chatting together as a group. With three of them based elsewhere (well happily Olivia is back to stay now) seeing them mingling and joking together really made my day. I know what it's like being an outsider or being based in a different location to the rest of the group so I was really pleased to see them integrated as a team. We unsurprisingly had a nice evening first sitting in the warm spring sun, smelling the cut grass and laughing about the noise pheasants make, and then enjoying some good pub grub (that's an Aussie term for food). Friday morning our team first years were all presenting in the same session (conveniently for me!). I remember how scary it is to give the first big seminar. Mine was my honours seminar at the University of Queensland (sort of like masters in some other countries). I was nervous as anything, stomach churning. I started ok, then disaster struck! I flicked to a slide and went completely blank. There was a graph on the slide but it might as well have been a fully black slide. I stared blankly for what felt like hours (probably 10 seconds). I was convinced I'd completely messed it up. This fear of messing up came back to haunt me when I worked in forestry research and had to present a seminar to industry funders. With my honours blank-out fresh in mind I was terrified. About 10 minutes before I was due to present, my boss lent over and whispered to me 'no pressure, but the next $20K depends on this' (I hadn't quite worked out yet that my boss was a tease - thanks Mark!). After that I honestly thought I would pass out. I still don't know how I got through that talk. It still makes me feel a little bit ill thinking about that experience and I confess, it all came flooding back as I nervously waited for the postgrad session to start on Friday morning. I was also worried they hadn't had enough support in practising, since I'd been waiting for an emergency hospital appointment the one day we all had to practice. I had made time to go through slides with each of them but it's not the same as standing up and practising the order of words. Reflecting back, I had no doubt they can all present well, instead my nerves were all about hoping they wouldn't develop a fear as I had! Remembering how valuable nodders and smilers are in the audience I chose a seat right in the middle close to the front to actively be that person for them. After all, the hard work in preparing is over by this stage so no need to scrutinise - that's what the assessors will do! The whole session was excellent. Rumour had it, that was one of the best sessions of the meeting (not my words, although I do believe it - but then more than half the talks were by students in my team - nothing unconscious about that bias!). This rumour however, was supported by the fact that both the gold and silver talks for our division came from that session - and since my guys really are the best, gold and silver went to Daisy Dobrijevic and Darwin Hickman! Although I'm super excited that it went to people in our team, full credit goes to their hard work. And even though only a few can receive awards, all of my students worked hard, improved their slides and gave clear talks (and chaired sessions like a professional!), and those not presenting went along and supported the others. As a supervisor I can't ask for anything more than that! Well done everyone! :) This week has been a challenge for me as an Australian living in the UK, watching the cricket scandal unravel. First comes the shock and disbelief, followed quickly by utter soul crushing disappointment and shame at being Australian.
Too extreme? You can learn more about what cricket means to Australia in this article. I could joke that the level of reaction is because our national pass-time is to sledge others who cheat, so now what are we to do? Seriously though, for me the parallels to academia are what sadden me even more deeply than the shame shared by a culture and I think there are some warnings and lessons that can be gained from this week's cricket scandal. What exactly do I mean? Like in cricket, where the leadership has led to a culture of less than honourable behaviours and extreme fatigue in the team, in academia the pressure put on individuals by various levels of management is taking its toll. Morale in academia is at an all-time low (see links at end of article). Is it any wonder that falsifying data to get high impact papers (akin to ball tampering to get better spin) is far from abating? On the one hand we need a work-life balance and on the other, each individual must achieve high ranking papers, pull in large grants in the face of ever decreasing funding sources, be leaders in our own right to our teams and receive high teaching scores from our undergraduate courses - and we must achieve this all of the time. Is it any wonder that stress leave is on the rise? How long before the academic work force crumbles like the Australian cricket team? But what would actually improve the academic culture? As someone on the lowest level of the academic ladder what I suspect would be the most important change is a return to team outputs. Instead of individuals being forced to spend time on activities for which they are less suited (and usually enjoy less), they could be encouraged to maximise the aspects of the job for which they are stronger (and usually enjoy more). The team as a whole would be much stronger and morale would undoubtedly improve. A second change is the inclusion of staff in decision making. Had the Australian cricket team been consulted as a team on the decisions being made about ball tampering, I'm certain it would not have happened (at least in my naivety that's what I'd like to believe). If management decisions include open and timely discussions with the people affected, more options will become available to decision makers, those affected will feel more in control of their surrounds, feel more valued by management and less upset by decisions that may still prove disruptive (see TED talk links below) The optimist in me would like to think there is hope. The Australian Cricket team will bounce back. The question is, without a nation in public uproar is there enough momentum to improve academia? Additional links: http://www.espn.co.uk/cricket/story/_/id/22928667/australia-stumble-cricket-moral-maze-reverse-swing University morale and depression rates https://www.theguardian.com/education/2017/may/04/ucl-staff-morale-at-all-time-low-because-of-management-poll-finds-academic-business-university http://www.bbc.co.uk/news/uk-wales-mid-wales-31946666 https://www.timeshighereducation.com/news/academics-face-higher-mental-health-risk-than-other-professions https://www.chronicle.com/article/The-Top-5-Faculty-Morale/236231 https://ebrary.net/2849/management/causes_morale TED talks on Leadership Radical transparency: https://www.ted.com/talks/ray_dalio_how_to_build_a_company_where_the_best_ideas_win/transcript https://www.ted.com/talks/simon_sinek_why_good_leaders_make_you_feel_safe/transcript Inclusive Leadership: https://www.ted.com/talks/jim_hemerling_5_ways_to_lead_in_an_era_of_constant_change/transcript Fairness: https://www.ted.com/talks/marco_alvera_the_surprising_ingredient_that_makes_businesses_work_better/transcript AR_lab may now have quite a few members but until last week most of them had not met each other! With Olivia in Adelaide, Erica in Brisbane, Magda in East Malling and Darwin in Rothamsted and with Findi in her first week getting together has been challenge! But last week we managed! - sort of! From left to right: Magda, Daisy, Darwin, Findi; on computer Erica (left) and Olivia (right) and me (in the tiny inset screen bottom right) Magda and Darwin made the journey to Nottingham and Erica and Olivia skyped in meaning we covered 3 time zones, two hemispheres and two very different seasons (with a layer of snow here that day!). We are also from 5 different countries including Spain, UK, Nigeria, America and Australia (with an American and Brit living in Australia and the Australian living in the UK!). The lab meeting was based around a 5-minute-thesis style talk (no powerpoint, something about ourselves and something about our research) followed by a chat about our different and complimentary skills sets. Then the fun stuff! Together we braved the weather (and traffic) and the 5 of us on site, piled into a car and headed to Calke Abbey (normally about 20 minutes from campus). I feel the title for a book coming on: Five go on a team building day! We had fun exploring the gardens and seeing the snowdrops despite the cold weather! Here are a few pictures from our afternoon... When we got back to campus we all gathered in the tea room for a nice hot cuppa before train journeys home!
I have to say I'm incredibly excited about this group of people. Fun, bright and motivated! Watch out world...my model team are on the way up! As we bring 2017 to a close, I wanted to reflect on positive things from the past year which has been filled with many challenges both personal and professional and I want to thank the people who have brought joy, support and inspiration. This year has seen me move division to Agriculture and Environmental Science (AES), has seen my team grow, teaching increase, new collaborations and strengthening existing collaborations, brought a range of amazing speakers to Biosciences, and seen a new Vice Chancellor start at the University of Nottingham. I first wish to thank everyone in AES for welcoming me into their community this year. They not only include me in research or teaching discussions but have also offered support and mentoring during some very challenging times. I can't speak highly enough of this diverse group of people. Elevenses brings social coffee time with the "tub of love" (a biscuit-filled tin) for any students, postdocs, technicians and academics around each day without any hierarchical division and usually ends in tears of laughter. Adding to AES, my team has grown welcoming Daisy Dobrijevic and Magda Cobo Medina, along with Darwin Hickman as a co-supervised student. In the coming year Findi Ishaya will join us from Nigeria on a VC PhD scholarship and we will host to Erica Porter from the Queensland University of Technology for a part of her Mphil. I have to say these wonderful people fill me with excitement with their positive attitudes and creative ideas. Exciting year to come! In addition to the growth of AR_Lab, my teaching portfolio is also growing. This calendar year I had the joy of teaching some of my favourite subjects in plant ecophysiology to 3rd years and running a new plant prac to about 300 first year students on two separate campuses. The discussions I had with some of this cohort while we wandered around trees in the glorious spring sun were of such a high standard that for 2018 I'm adding a third stage to the prac involving a treasure hunt using a mobile phone app. This makes me a tad nervous regarding technological glitches so I have a (dreaded) paper backup for the first run but I'm also really excited to be able to stretch the students further in their plant evolution and diversity knowledge in a fun way. The autumn semester saw the beginning of a new academic year with a delightful group of 4 first year tutees and saw last years' tutees move on to internship applications and a higher level of challenges and time management. My five 3rd year project students have embarked on some really diverse and interesting projects from coastal erosion management to moss invertebrate diversity to crop nutrient physiology (in response hydroponics/soil and in response to pharmaceuticals present in wastewater) and to nitrogen preferences of plants growing in different niches (potentially interesting for greenspace in urban areas). I taught into two plant physiology modules at 2nd year and masters levels and received some truly wonderful comments on my teaching scores (if you follow me on twitter you will have seen my surprised reaction!). I have to say my job is made easy when the room is filled with motivated, interactive students so thank all of you who were in my classes this year. The year also involved 3 trips to Sweden to visit friends, collaborators, to attend the rooting 2017 conference (see blog and here) and to take 20 undergraduate students to the arctic for a field module (see blog). Collaboratively, 2017 saw several international peers start in academic positions (and I signed my permanent contract too). One of those amazing people is Erin Sparks (Dellaware) and together we set up a sister-lab arrangement to collaborate on maize root-type research which is incredibly exciting - both in terms of research but also in terms of developing a peer group who understand the challenges of being a new academic in current times. Many other collaborators in Germany, Australia, Sweden, Netherlands, Italy and here in the UK have also been amazing for feedback and support with grant applications over the past year. This includes support from Karl Pioch at KWS SAAT SE and from Kevin Hobbs at Hillier Nurseries as we develop new industry-relevant project. Very recently I had the pleasure of attending the Hillier Lancaster Plant Group and meet Roy Lancaster and a room full of other horticulture buffs. This was an amazing and humbling experience being surrounded by so much plant knowledge - a resource I plan to include in my teaching a research more in coming years! One of my roles earlier this year was to organise the Holden Botany public Lecture and organise the seminar series. As part of this we hosted some incredible speakers including Sandy Knapp from the Natural History Museum (Holden Botany Lecture), Ottoline Leyser, Levy Yant, Ari Sadanandom, Katie Field and Tom Bennett (seminar series). In addition just this week I had the great pleasure of meeting James Wong (@botanygeek) who visited Nottingham to give a public seminar organised by Susie Lydon which raised money for UNICEF. It was a fascinating talk about how we can use a knowledge of plant processes (AKA physiology!) to improve the nutritional value of fruit and vegetables in our homes. Hearing from these inspirational people is one of the job perks of being an academic and I thank them all for taking the time to visit the University of Nottingham. So at this point I want to thank you all - AR_Lab members, students, colleagues, collaborators, and friends for the good things this year and I look forward to exciting times to come in 2018. The year has also seen the University of Nottingham welcome a new Vice Chancellor to our ranks. I have just watched Prof. Shearer West's inaugural lecture and I have to say I feel more optimistic about the future.
In a world filled with awareness for unconscious bias I have been reflecting on why I find Prof West an inspiring leader…*disclaimer: in the following statements I am by no means suggesting I am anywhere near the same league as Shearer but rather attempting to dissect potential biases of mine* … So do I find her inspirational because she's a woman? Or because she's international? Or is it because she's not afraid to tackle problems and give opinions that are not necessarily going to be 'liked' by everyone? Is it because she made a strong case for supporting early career academics to be able to work with the best in our field? Is it because she drew on expertise and quotes from VCs of several Australian institutions? I happen to agree with many of the things she said, but I agree with many people and don't necessarily find them inspirational. I think what makes her an inspiration to me are those unconscious biases - she says things I relate to and agree with but she does it as an international woman in a position of power. But what does this mean for me as I improve my own leadership skills for my team? AR_Lab is internationally diverse including English, Spanish, Nigerian and American/Australian (and me as an Australian). Interestingly almost all PhD student applicants have been female - (that's before I get any say in the process!). At postdoc level it is much more balanced and together with Darwin joining the team I'm pleased that we are becoming more diverse on that front. We are still small and new so the true diversity of experiences, opinions and personalities are yet to fully shine but on reflection I think my fledgling team is growing well in terms of diversity. The real challenge now will be to find a way to inspire people diverse from myself; to establish a set of common goals with each of them to inspire them through 'the cloud' times (see blog…) and encourage a positive team environment to give them resilience to the challenges (including frustrating supervisor moments)! I am new to leading a team and I'm certain I have much to learn and will make plenty of mistakes but hopefully as a team we can evolve together. Well that turned into quite the reflective piece! But what better time to take stock and reflect than at the end of a calendar year! Reflecting back on the amazing things that have happened in the past year and being motivated by my research team and undergrad students, I am looking forward to 2018 with excited anticipation. Happy holidays everyone. Hi Everyone! It's been a while since I posted...been busy...you know the drill (and all the excuses!). Part of the being busy this time is that one of my new procrastination activities is being involved in the Plantae network/platform. This is a platform for anyone interested in plants to post advice, ask questions, read articles - basically interact on a global scale. It's like facebook for planty people. This includes career advice and job adverts. It's free to sign up (and you can be as anonymous as you like if you choose to use a different user name). Details in the flyer below. I want to emphasise that ANYONE can sign up - there are networks for teaching, for different plant societies and articles and advice on all types of plant science. It's getting momentum with some great interactions and resources available which is great - so come and join us if you haven't already! Personally I'd also love to see more schools/school teachers sign up and to help this become a bridge between school and university plant science teaching (and if you are a teacher and have questions, feel free to email me - we are still exploring what this platform can do and your advice and questions are very welcome). As an example I just posted a comment on mentoring in response to a fabulous article written by Mary Williams on getting the mentoring you need. You can read Mary's article here but to comment you will need to sign up. I've posted my comment below the advert. "I always find this [mentoring] an interesting topic. The advice above [in Mary's article] is really good. Although I didn’t have it put so plainly, this is similar to advice I collated over my early years at Uni. The most important message in all of this is that it’s up to you to find mentoring that works for you – academia is incredibly diverse with different people having different requirements and ultimately it’s your life so make the most of it!
I think a key thing to remember is that there are different types of mentor. There are the official ones allocated to you by the institution, there are unofficial mentors you create (in my experience a lot of this is dependent on personality types) and there are your friends. I’m going to just briefly comment on each type and my experiences. The official mentors: these are people allocated to you by your institution and different institutions have different allocation strategies with one common theme – none of them automatically work for everyone. At Nottingham when I first arrived I was given a questionnaire with a range of questions, most of which I’ve forgotten – the most important one was “do you want someone in your division?”. My answer was an emphatic NO! For me I needed someone who I could ask advice outside the divisional structure and outside my own line management structure (at least a little bit). And that was probably the first really good strategic decision I made here. That person is still a mentor (probably still officially) and will remain a mentor for life. A lot of people at Nottingham think the official mentoring scheme doesn’t work but I think it’s up to us to make it work for us. To mentors – I think it’s particularly important to take new staff under our wings – they are the ones who don’t know who to approach at the beginning, and the beginning is the steepest learning curve (BELIEVE ME!). Unofficial mentors: I have a lot of these and they fall within the different categories in Mary’s article above. Some are likely to be on my promotions panel one day, some are colleagues who are a few years ahead of me and have been through the early career academic years recently enough to provide helpful advice on things from university structures to coping with the onslaught of email requests. Some are people who I’ve met along the way. I have several mentors in Australia where I studied and where I worked in industry and I regularly call on them for advice (always when I’m back in Aus but also from time to time from the UK). I recently reconnected with two different mentors who I haven’t been in touch with since before I left Australia almost 6 years ago. One of them I haven’t seen since my honours year finished back in 2005. I’ve always found people like to find out where I’ve ended up (and I think that’s regardless of where I could have ended up). So the advice about staying in touch with mentors is really important! Friends: This group is perhaps the most important group of mentors and probably the least talked about. This doesn’t mean every friend is a mentor – this is the group of people that you go to in times of difficulty. Your friends are the people you can talk to about any problem including those linked to work. They should be able to give you comfort for difficult situations and an airing board for possible solutions before you go and talk to your unofficial or official mentors. If you’re like me and get really frustrated with certain situations, the friend-mentors are the people I rant to (usually with a heated voice, often with tears) until I calm down and can approach a problem more strategically (those of you who know about myers-briggs – I’m an E(I)NFJ –I sit on the E-I divider and F(feeling) and J(lists-about-lists) are my stronger traits). The friends-mentors group is the group I missed a lot when I first moved here – I still had my friends elsewhere but they weren’t here and crying at a computer screen over skype doesn’t have the same effect as someone sitting in the same room (those of you going through that – I get it totally). It takes time to build that group of friend-mentors and I have learnt to be persistent with skype because it does take the edge off even when I’m in my introvert mode. I have found that being on a Nottingham Research Fellowship has helped this because some of the other fellows have become strong friends and we take time for coffees now and then when I’m at our University Park campus. This is what my experiences have been but I really think mentoring is up to YOU to find what YOU need. Be persistent in asking for what you need and be pro-active within your institution (or wider community) – the best mentors are the ones you choose through interaction, whether that’s through going to seminars, or professional development courses or on the university tennis team. I’m going to end there and just remind everyone to not only look up for mentors but to pay it forward – I had people help me, so I feel it's my duty to mentor others." |
AuthorAmanda Rasmussen Archives
May 2023
Categories |























 RSS Feed
RSS Feed
